What Is FINASTERIDE 5MG/ML | 60ML?
Finasteride, sold under the brand name Proscar, is a 5α-reductase inhibitor. It is an antiandrogen FDA-approved drug used for the treatment of benign prostatic hyperplasia and male pattern baldness. It has also been shown to be effective in the treatment of hirsutism. Furthermore, researchers are exploring its potential effect on sleep apnea and central serous chorioretinopathy. At Pinnacle Peptides, finasteride for sale is exclusively available for research and experimentation.
Structure Of FINASTERIDE 5MG/ML | 60ML
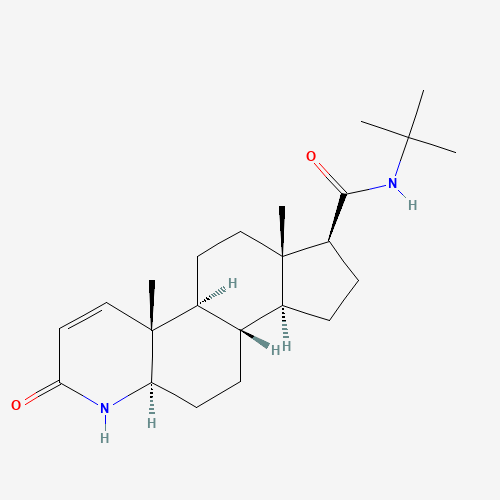
From Pubchem
IUPAC Name: (1S,3aS,3bS,5aR,9aR,9bS,11aS)-N-tert-butyl-9a,11a-dimethyl-7-oxo-1,2,3,3a,3b,4,5,5a,6,9b,10,11-dodecahydroindeno[5,4-f]quinoline-1-carboxamide
Synonyms: Proscar, Propecia, Finastid
Molecular Formula: C23H36N2O2
Molecular Weight: 372.5 g/mol
CAS number: 98319-26-7
PubChem CID: 57363
History
A researcher named Imperato-McGinley described a group of male children lacking 5α reductase enzyme in 1974. All of them exhibited low levels of DHT and their postrates remained small throughout their lifetime. This led to the discovery of a drug, finasteride, that could inhibit the 5α reductase enzyme. The drug was patented in 1984 and was approved by the FDA for the treatment of benign prostatic hyperplasia in 1992. Five years later, the FDA also approved it for the treatment of male pattern baldness in 1997.
Mechanism Of FINASTERIDE 5MG/ML | 60ML
Finasteride is a competitive inhibitor of type II and III 5α reductase inhibitor, an enzyme responsible for the conversion of testosterone to dihydrotestosterone (DHT). High levels of DHT are associated with the maturation of follicles and enlargement of the prostate gland.
Literature shows that finasteride has a minimal selectivity towards type I 5α reductase enzyme which is present in sebaceous glands, sweat glands and epidermal keratinocytes. On the other hand, type II 5α reductase is found in the outer sheath of hair follicles, seminal vesicles and prostate glands. Research has shown that finasteride can lower serum DHT levels by 70% and DHTlevels in the prostrate by 90% [1].
Pre-Clinical/Clinical Research
1. Androgenetic Alopecia
Finasteride has been shown to be effective against androgenetic alopecia. Literature suggests this drug improves hair loss by 30% within six months of therapy [2]. Although it is effective, its systemic use causes various side effects, such as gynecomastia, decreased ejaculate volume, male infertility and high-grade prostate cancer. This limits the long-term utilization of the medicine.
Alternatively, topical finasteride show better results for androgenetic alopecia with more favorable side effect profile than systemic therapy. A systemic review concludes that topical finasteride decreases the rate of hair loss and increases total and terminal hair count. Furthermore, it has been shown to effectively reduce the concentration of DHT in both scalp and plasma [3].
Finasteride has also been explored for the female pattern baldness but it didn’t produce any significant results [4]. Moreover, one study was conducted to look at the effect of 5α reductase enzyme inhibitor, finasteride, on androgenic alopecia in female-to-male transgender patients. All the patients who took one 1 mg oral finasteride improved one grade on the Norwood-Hamilton scale after 4-6 months of initiating therapy [5].
2. Central Serous Chorioretinopathy
Central serous Chorioretinopathy (CSC) is an idiopathic disorder characterized by the accumulation of fluid behind the retina causing serous detachment of the macula, resulting in impaired vision. It has been proposed that elevated testosterone levels are associated with the pathogenesis of CSC. Research suggests that 5α reductase enzyme inhibitor, which prevents the conversion of testosterone into dihydrotestosterone, might be effective for treating CSC [6].
One study was designed to evaluate the safety and effectiveness of finasteride in patients with CSC. It found that patients who took finasteride for CSC reported improvement in visual activity. Furthermore, a reduction in macular thickness and subretinal fluid was observed, suggesting the efficacy of finasteride in treating this disorder [7].
3. Prostate cancer
Androgens play a key role in the pathophysiology of prostate cancer. 5α reductase inhibitors, including finasteride, prevent the formation of primary androgen (dihydrotestosterone) in prostate tissues. They might prove effective in treating or preventing prostate cancer.
A study named Prostate Cancer Prevention Trial (PCPT) was designed to study the effect of finasteride on prostate cancer. Almost 19,000 men aged 50 or older having PSA levels of 3ng or lower were enrolled. The subjects were randomly assigned to receive either 5 mg finasteride or an equivalent placebo once a day for seven days. The researchers found that finasteride delays or prevents the occurrence of prostate cancer [8].
Interestingly, it was observed that high-grade tumors were more prevalent in the treatment group as compared to the placebo group [8]. Later, it was suggested that it might be due to finasteride’s impact on prostate volume and its potential to suppress low-grade cancer rather than its effect on tumor morphology. Furthermore, the high-grade tumors were detected earlier and were less extensive in the treatment group as compared to the control group [9].
4. Hirsutism
Hirsutism is a condition characterized by excessive growth of the body and facial hairs. It is often associated with high levels of androgens. Finasteride may benefit people with hirsutism. A study was conducted to check the impact of 5α-reductase inhibitors on hirsutism in women with PCOS. Subjects enrolled in the study were administered with a placebo or finasteride once a day for seven months. The results found that finasteride produced a reduction in hirsutism score and was found to be well tolerated and effective [10].
Summary
Finasteride competitively inhibits 5α-reductase, an enzyme responsible for the conversion of testosterone into dihydrotestosterone. It possesses selectivity and doesn’t affect adrenal synthesis. It is approved by the FDA for the treatment of benign prostatic hyperplasia and alopecia. Furthermore, researchers show that it can reduce the incidence of prostate cancer and might treat central serous chorioretinopathy. We don’t support its unwarranted use and offer finasteride purchase solely for research. Only buy finasteride if you are a qualified researcher.
References
- Zito PM, Bistas KG, Syed K. Finasteride. [Updated 2022 Aug 25]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-.
- Varothai, S. and W.F. Bergfeld, Androgenetic Alopecia: An Evidence-Based Treatment Update. American Journal of Clinical Dermatology, 2014. 15(3): p. 217-230.
- Lee, S.W., et al., A Systematic Review of Topical Finasteride in the Treatment of Androgenetic Alopecia in Men and Women. J Drugs Dermatol, 2018. 17(4): p. 457-463.
- Levy, L.L. and J.J. Emer, Female pattern alopecia: current perspectives. Int J Womens Health, 2013. 5: p. 541-56.
- Moreno‐Arrones, O.M., A. Becerra, and S. Vano‐Galvan, Therapeutic experience with oral finasteride for androgenetic alopecia in female‐to‐male transgender patients. Clinical and Experimental Dermatology, 2017. 42(7): p. 743-748.
- Forooghian, F., et al., Finasteride for chronic central serous chorioretinopathy. Retina, 2011. 31(4): p. 766-71.
- Moisseiev, E., et al., Finasteride is effective for the treatment of central serous chorioretinopathy. Eye, 2016. 30(6): p. 850-856.
- Thompson, I.M., et al., The Influence of Finasteride on the Development of Prostate Cancer. New England Journal of Medicine, 2003. 349(3): p. 215-224.
- Lucia, M.S., et al., Finasteride and High-Grade Prostate Cancer in the Prostate Cancer Prevention Trial. JNCI: Journal of the National Cancer Institute, 2007. 99(18): p. 1375-1383.
- Tolino, A., et al., Finasteride in the treatment of hirsutism: new therapeutic perspectives. Fertil Steril, 1996. 66(1): p. 61-5.
What is Finasteride?
FINASTERIDE is a type II isoform of 5-alpha reductase. It is an anti-androgen, which means that it converts testosterone into DHT. This peptide has been deemed useful in treating benign prostate hyperplasia or BPH. If taken in higher doses, it has also been seen to be effective in treating prostate cancer patients. This product primarily treats two conditions: male pattern hair loss and benign prostatic hyperplasia (BPH). Finasteride was initially created to treat BPH, but it was later found to promote hair growth. It prevents the enzyme 5-alpha-reductase from converting testosterone into the hormone dihydrotestosterone (DHT), which is linked to hair loss and enlargement of the prostate. Finasteride can reduce hair loss and encourage hair regrowth in males with male-pattern baldness by lowering DHT levels. Finasteride reduces symptoms of BPH by shrinking the prostate gland. Currently, research has been going on to study the other beneficial effects of this peptide.
Finasteride Research
Male pattern hair loss (androgenetic alopecia) and benign prostatic hyperplasia (BPH) can be treated with the drug finasteride, which has undergone significant investigation and is frequently prescribed. Here, we'll examine the major discoveries and developments in finasteride research, illuminating its workings, efficacy, and potential negative effects.
Mechanism of Action:
According to research, Finasteride's therapeutic benefits are believed to be mediated through inhibition of the enzyme 5-alpha-reductase, especially the type II isoform. Dihydrotestosterone (DHT), an essential hormone for hair loss and prostate enlargement, is created when this enzyme transforms testosterone. Finasteride decreases DHT levels by preventing the conversion of testosterone to DHT, which slows hair loss and shrinks the prostate gland.
Effectiveness in Male Pattern Hair Loss:
Clinical studies have shown that Finasteride effectively treats male pattern hair loss. Finasteride considerably enhanced hair growth and lowered hair loss compared to a placebo, according to a landmark study involving more than 1,500 males. The vertex (crown) and the anterior mid-scalp regions showed this improvement. Long-term research has further supported its pervasive effectiveness, with continuous hair maintenance and growth even after extensive use.
Efficacy in Benign Prostatic Hyperplasia:
Finasteride has been proven effective in treating BPH through extensive study. According to studies, Finasteride lowers prostate volume, enhances urinary symptoms, and boosts peak urine flow rates. It is a helpful BPH therapy option because long-term studies have shown that it can maintain these benefits over time.
Side Effects and Safety Profile:
Research has highlighted potential side effects associated with finasteride use. While generally well-tolerated, some people may develop adverse sexual effects like decreased libido, erectile dysfunction, or ejaculation issues. Usually, these side effects go away after the medicine is stopped. Finasteride use has sporadically been linked to sadness and anxiety, but further studies are required to draw a firm conclusion. Healthcare providers must educate patients about potential side effects and monitor how they respond to treatment.
Where to Buy Finasteride
At Pinnacle Peptides, you can buy Finasteride online in the purest form possible. You must be careful when purchasing these products since most provide impure or diluted ones. But Pinnacle Peptides understands your need for pure peptides to receive accurate results. When purchasing FINASTERIDE, trust Pinnacle Peptides to provide you with the purest forms. If you require the peptide for your experiment, you can buy the vials as per your requirement and place the order on the website. We provide you with a wide range of peptides for your research. You will surely get quite a deal with quality products and the best prices when you shop online.
Disclaimer
The products we offer are intended for laboratory research use only. In purchasing any of these items, the customer acknowledges the risks of consuming or distributing these products. These chemicals are NOT intended to use as food additives, drugs, cosmetics, household chemicals, or other inappropriate applications. Listing a material on this site does not constitute a license to its use in infringement of any patent. The products will be handled only by qualified and properly trained professionals. All customers represent and warrant that through their review and study, they are fully aware and knowledgeable about the following: Government regulations regarding the use of and exposure to all products—the health and safety hazards associated with handling the products they purchase. An adequate warning of any products' health and safety hazards is necessary. Pinnacle Peptides reserves the right to limit and deny sales of products to any unqualified individuals if we have reason to believe that misuse will occur.