What Is LIQUILETRO 3.5MG / ML | 60ML?
Letrozole is a third-generational non-steroidal aromatase inhibitor. It is an FDA-approved drug primarily indicated for the treatment of breast cancer in postmenopausal women. It has also been prescribed for ovulation induction in patients with PCOS. Furthermore, research shows that it can improve clinical response to endometrial cancer. Currently, it is undergoing clinical trials for its efficacy in hepatopulmonary syndrome. At Pinnacle Peptides, letrozole for sale is exclusively available for research and experimentation.
Structure Of LIQUILETRO 3.5MG / ML | 60ML
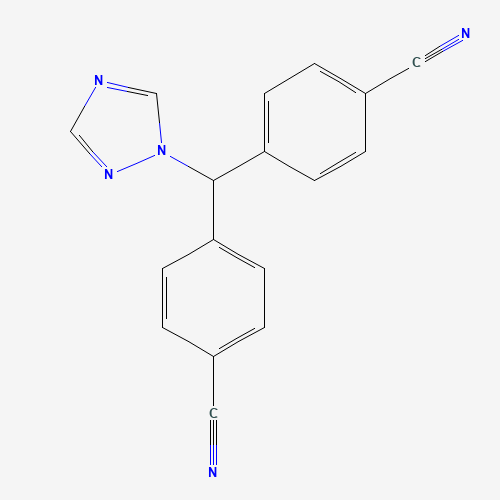
From Pubchem
IUPAC Name: 4-[(4-cyanophenyl)-(1,2,4-triazol-1-yl)methyl]benzonitrile
Synonyms: 112809-51-5, Femara
Molecular Formula: C17H11N5
Molecular Weight: 285.30 g/mol
CAS number: 112809-51-5
PubChem CID: 3902
History
Letrozole was first discovered by the Swiss company Ciba-Geigy AG in the 1980s in order to develop a drug that could inhibit the aromatase enzyme. The researchers found letrozole to be more potent than other drugs from the same class. This led to its accelerated development, resulting in its approval from the FDA in 1996 for the treatment of advanced breast cancer. In 2004, the FDA expanded its approval for adjuvant treatment.
Mechanism Of LIQUILETRO 3.5MG / ML | 60ML
Letrozole competitively inhibits the active site of the aromatase enzyme. Aromatase is a member of the CP450 superfamily monooxygenases enzyme. It catalyzes the demethylation of carbon 19 of androgen, resulting in the synthesis of estrogen. In postmenopausal women, aromatase is responsible for the major source of estrogen production in peripheral tissues, ovaries and the brain. By blocking the activity of aromatase, letrozole reduces the concentration of hormone, which reduces estrogen-mediated proliferation of cancer cells. Furthermore, low levels of estrogen in ovarian follicles send a signal to the hypothalamus to release gonadotropin-releasing hormone (GnRH). This, in turn, stimulates the ovaries to produce FSH and LH, leading to ovulation [1].
Pre-Clinical/Clinical Research
1. Endometrial Cancer
Research shows that endometrial cancer can be cured with traditional therapy and surgery in the majority of the patients. However, stats reveal that 8000 patients die annually because of the resistance to treatment. Literature indicates the association between the PI3K/AKT/mTOR signaling pathway and the progression and unfavorable prognosis of this disease. Additionally, estrogen receptor signaling contributes to the development of type 1 endometrial cancer and may boost the expression of proteins responsible for activating the PI3K/AKT/mTOR pathway.
One study showed that a combination of PI3K/AKT/mTOR pathway inhibitors and hormone therapy might hold promising results for individuals resistant to treatment. It is expected to yield a synergistic effect, potentially enhancing the response rates in patients with endometrial cancer. A phase II study specifically observed that the combination of letrozole and everolimus (PI3/AKT/mTOR inhibitor) significantly improved the clinical benefit ratio and response rates among women suffering from recurrent endometrial cancer [1].
2. Infertility
There is extensive research that proves the efficacy of letrozole in treating infertility. A meta-analysis compared the effect of eight different treatments for ovulation induction in women with anovulation. The result found that letrozole alone and the combination of clomiphene citrate and metformin have superior efficacy in terms of inducing pregnancy than other treatments. Furthermore, it found that letrozole treatment resulted in higher live birth rates as compared to clomiphene alone [2].
Furthermore, one study found that a combination of letrozole and clomiphene showed better results in inducing ovulation as compared to letrozole alone [3]. Research shows that letrozole along with GnRH therapy might prove helpful in preserving fertility in patients with endometrial cancer. One study found that it successfully enabled the retrieval of oocytes and embryo transfer, thus facilitating pregnancy [4].
3. Constitutional Delay in Growth and Puberty
Constitutional delay in growth and puberty (CDGP) is responsible for short stature that typically affects boys. Individuals with CDGP undergo a delay in the onset of pubertal growth. However, they catch up by the age of 18 during which their growth rate accelerates and they achieve normal height.
Children with CDGP often face psychosocial pressure, which makes the treatment important. Studies show that letrozole stimulates the hypothalamus-pituitary-gonad axis, which results in the quick growth of testicles and enhanced pubertal progression. Furthermore, it has been shown to be well tolerated and efficacious in improving testicular growth and delaying bone age progression as compared to testosterone [5].
4. Breast Cancer
Letrozole is widely prescribed to treat local and metastatic breast cancer in postmenopausal women with hormone receptor-positive tumors or of unknown receptor status. Still, some patients with early-stage breast cancer face a continuous risk of recurrence. The Breast International Group (BIG) 1-98 study found that letrozole monotherapy significantly decreased the risk of recurrence and mortality in postmenopausal women with hormone-sensitive breast cancer compared to tamoxifen. However, the sequential treatment didn't show any better outcomes than letrozole monotherapy, but it still improved the tolerability of treatment [6]. Sequential treatment consists of the use of one drug for two years followed by another drug for three years.
Research suggests that resistance to letrozole can develop over time in some patients, leading to decreased responsiveness of breast cancer cells to the drug. Animal studies suggest that intermittent letrozole therapy could reverse this resistance. The SOLE (Study of Letrozole Extension) clinical study revealed that the extended intermittent treatment (9 months of therapy followed by a 3-month break) for five years effectively improves patients’ quality of life. Furthermore, the results suggested that it is safe for patients undergoing letrozole therapy to take temporary breaks in between the treatments [7, 8].
Summary
Letrozole is a non-steroidal aromatase inhibitor that suppresses the conversion of androgens into estrogen. The low exposure to estrogen reduces the proliferation of hormone receptor-positive breast cancer. Furthermore, it stimulates the hypothalamus-pituitary-gonadotropin axis to induce ovulation. Research shows that it can treat endometrial cancer and optimize growth in children with CDGP. At Pinnacle Peptides, we offer the option to purchase letrozole exclusively for research. Only buy letrozole if you are a qualified researcher.
References
- Slomovitz, B.M., et al., Phase II study of everolimus and letrozole in patients with recurrent endometrial carcinoma. J Clin Oncol, 2015. 33(8): p. 930-6.
- Wang, R., et al., Treatment strategies for women with WHO group II anovulation: systematic review and network meta-analysis. Bmj, 2017. 356: p. j138.
- Mejia, R.B., et al., A randomized controlled trial of combination letrozole and clomiphene citrate or letrozole alone for ovulation induction in women with polycystic ovary syndrome. Fertil Steril, 2019. 111(3): p. 571-578.e1.
- Azim, A. and K. Oktay, Letrozole for ovulation induction and fertility preservation by embryo cryopreservation in young women with endometrial carcinoma. Fertility and Sterility, 2007. 88(3): p. 657-664.
- Dutta, D., et al., Efficacy and Safety of Letrozole in the Management of Constitutional Delay in Growth and Puberty: A Systematic Review and Meta-analysis. J Clin Res Pediatr Endocrinol, 2022. 14(2): p. 131-144.
- Regan, M.M., et al., Assessment of letrozole and tamoxifen alone and in sequence for postmenopausal women with steroid hormone receptor-positive breast cancer: the BIG 1-98 randomised clinical trial at 8·1 years median follow-up. Lancet Oncol, 2011. 12(12): p. 1101-8.
- Colleoni, M., et al., Extended adjuvant intermittent letrozole versus continuous letrozole in postmenopausal women with breast cancer (SOLE): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol, 2018. 19(1): p. 127-138.
- Ribi, K., et al., Quality of life under extended continuous versus intermittent adjuvant letrozole in lymph node-positive, early breast cancer patients: the SOLE randomised phase 3 trial. British Journal of Cancer, 2019. 120(10): p. 959-967.
-
I will recommendI will recommend LETROZOLE to everyone.
Posted on
-
I am very impressedI am very impressed. Love it.
Posted on
-
Order againI am going to order again soon.
Posted on
-
Good productAppropriate packaging and good product.
Posted on
-
This works greatI'm very satisfied with the purchased. The whole process went well. The product was wrapped well, and the shipping fast, and it really works. I will order again.
Posted on
-
It was my first timeIt was my first time ordering from this company and the process was quick and easy.
Posted on
-
Everything was as I orderedGood product, fast shipping, will purchase again
Posted on
-
Great prices!Delivery was fast.Prices were very competitive.
Posted on
-
Happy customer!Product received on time and in great condition :-)
Posted on
-
Very promptVery prompt
Posted on