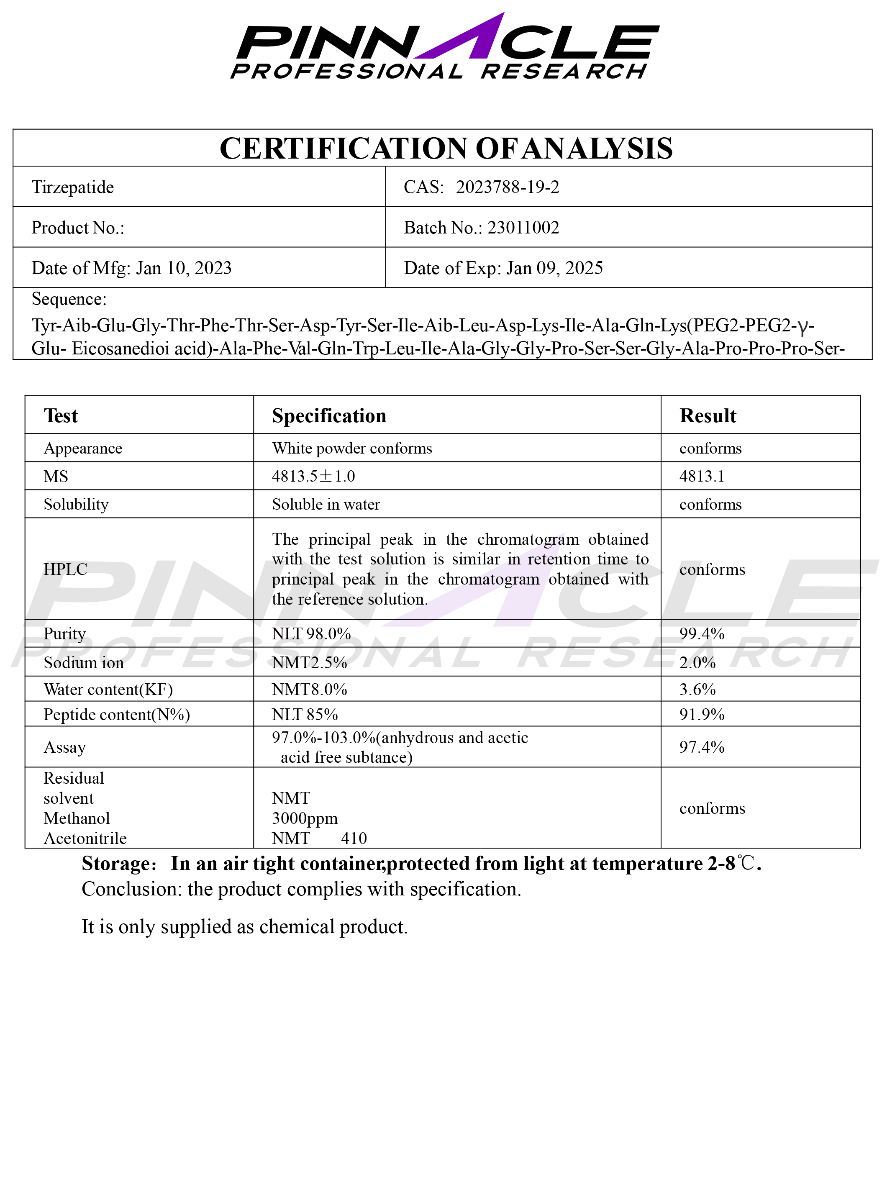
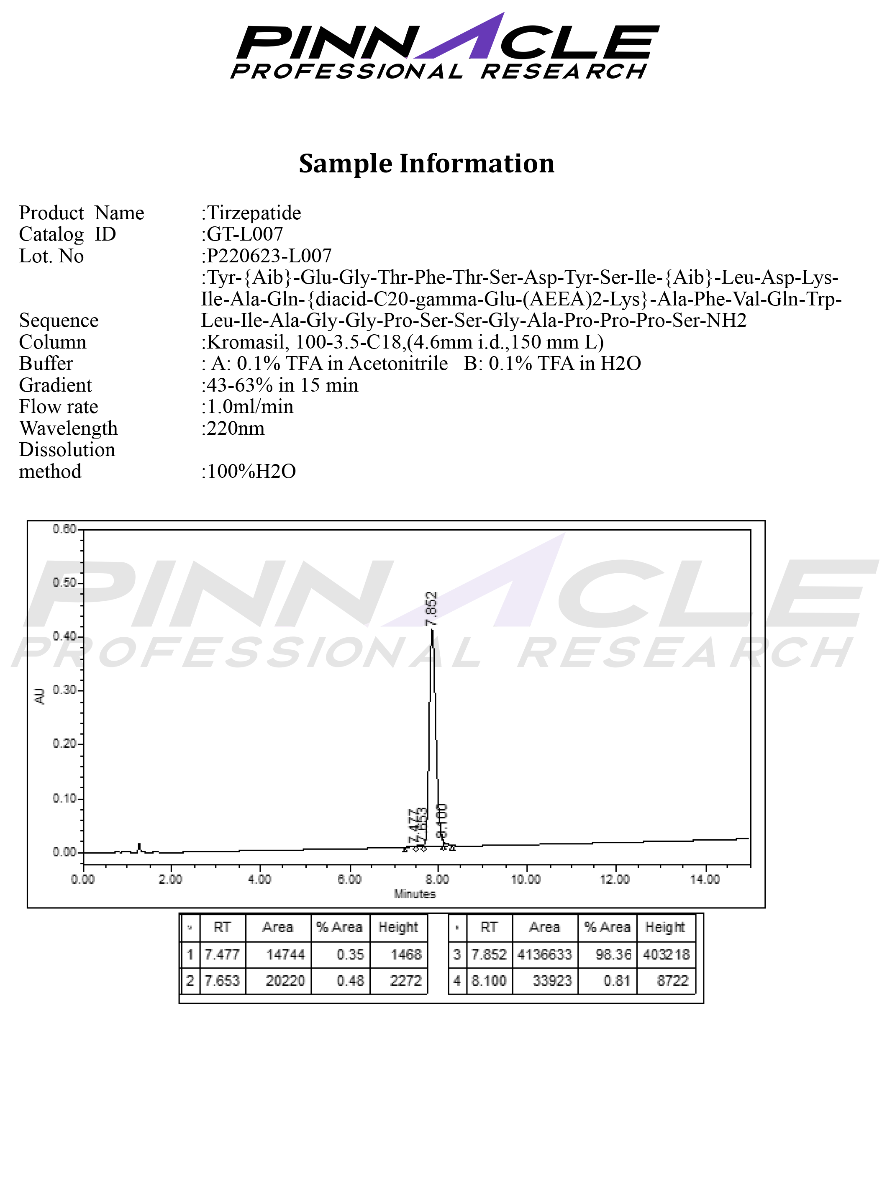
1. Cardioprotective Effects
Research shows that tirzepatide might offer cardioprotective effects. One study found that tirzepatide alleviated cardiac dysfunction induced by lipopolysaccharide and suppressed the inflammatory response in mice. Additionally, it reduced the risk of ventricular arrhythmia and cardiac apoptosis through inhibition of the TLR4/NF-kB/NLRP3 pathway [1].
A follow-up analysis of the SURPASS clinical program found that patients treated with TZP experienced a decrease in systolic blood pressure due to its impact on weight loss [2]. Furthermore, another study discovered that individuals who received TZP had greater reductions in triglyceride levels and LDL-C compared to those given insulin glargine [3]
2. Diabetes
The FDA approved Tirzepatide for type 2 diabetes following a series of successful phase 3 clinical trials conducted in the SURPASS program. These trials provided substantial evidence showcasing tirzepatide's superior effectiveness when compared to a placebo and other medications.
Results from the SURPASS 3 trials have demonstrated that Tirzepatide (TZP) outperforms Insulin Degludec in terms of efficacy. Notably, patients treated with TZP who achieved an HbA1c level of <5.7% over 52 weeks displayed more favorable outcomes in TIT, TTIT, and glycemic variability compared to those who did not reach this target [4].
Furthermore, another study indicates that when combined with insulin glargine, TZP leads to improved glycemic control in patients who were previously unresponsive to insulin glargine [5].
The SURPASS 6 trial has revealed that TZP is more effective in enhancing HbA1c levels and improving the health-related quality of life (HRQoL) in diabetic patients when compared to insulin lispro [6].
3. Weight Loss
The SURPASS 1-5 trials, which investigated the anti-diabetic effects of tirzepatide, have also demonstrated a significant impact of TZP on weight loss. A meta-analysis of these trials reveals that 43% of patients who received TZP lost more than 15 kilograms of body weight [7].
In the SURAMOUNT trial 1, researchers assessed the safety and effectiveness of tirzepatide in obese individuals. They enrolled 2539 participants in the study and randomly assigned them to receive subcutaneous TZP (at doses of 5mg, 10mg, or 15mg) or a placebo for 72 weeks. The findings indicated that the percentage of participants achieving more than a 5% reduction in weight was 85% for 5mg, 89% for 10mg, and 91% for 15mg. These stats clearly indicate TZP’s superior efficacy compared to the placebo [8].
Furthermore, SURAMOUNT trial 2 found that 79-83% of individuals treated with TZP experienced more than 5% weight loss, in contrast to the control group where only 32% achieved this level of weight reduction [9].