What Is LIQUITAMO 20MG / ML | 60ML ?
Tamoxifen is a non-steroidal selective estrogen receptor modulator (SERM). It has been shown to exert both agonist and antagonist activity at the estrogen receptors. Furthermore, it has been shown to activate heat shock protein. It is an FDA-approved drug prescribed for the treatment of breast cancer in men and women. It also has an off-label indication for the treatment of gynecomastia and infertility. Various studies indicate tamoxifen can increase muscle strength, leading to its ban by the World Anti-Doping Agency. Currently, it is being studied for its effect on myotubular myopathy and other types of cancers. At Pinnacle Peptides, tamoxifen for sale is exclusively available for research and experimentation.
Structure Of LIQUITAMO 20MG / ML | 60ML
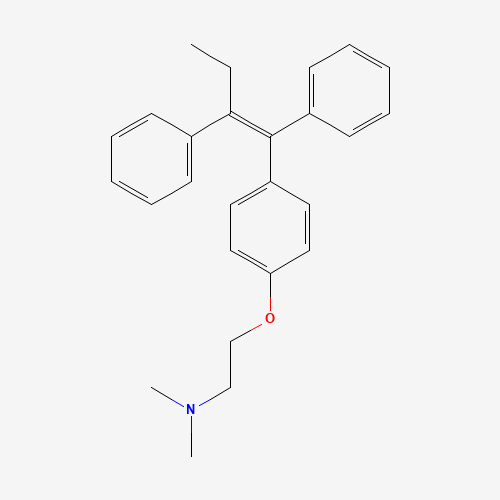
From Pubchem
IUPAC Name: 2-[4-[(Z)-1,2-diphenylbut-1-enyl]phenoxy]-N,N-dimethylethanamine
Synonyms: trans-Tamoxifen, Crisafeno, Diemon
Molecular Formula: C26H29NO
Molecular Weight: 371.5 g/mol
CAS number: 10540-29-1
PubChem CID: 2733526
History
It was first developed by a chemist named Dora Richardson in 1962 when she was trying to discover a compound that could work as a birth pill. Later, the researcher discovered that it induces ovulation instead of preventing pregnancy. Initially, it was approved for the treatment of fertility but it never proved useful in human contraception.
Back then, another scientist named Walpole convinced the company he was working with to test the drug for its effect on breast cancer. In 1971, the first human clinical study on tamoxifen showed promising results, leading to its development for the treatment of breast cancer. Ultimately, it was first approved by the FDA for advanced breast cancer treatment in 1977.
Mechanism Of LIQUITAMO 20MG / ML | 60ML
Tamoxifen is an estrogen receptor modulator and it has been shown to exhibit both estrogenic and antiestrogenic activity. It acts as an antagonist in the breast tissues and competitively binds to the estrogen receptor (ER). By blocking the hormone activity, it prevents ER binding to estrogen-response elements on DNA. This, in turn, slows down the cellular response to estrogen and decelerates the progression of the cell cycle.
In the bone tissues, it acts as an agonist of estrogen receptors. Instead of blocking estrogen receptors, it activates them and might prevent osteoporosis through this mechanism. Furthermore, it has been shown to exhibit agonist activity at the estrogen receptors present in the hypothalamus of premenopausal women, thus facilitating ovulation [1].
Pre-Clinical/Clinical Research
1. X-linked Myotubular Myopathy
Myotubular myopathy, also known as X-linked myotubular myopathy (XLMTM), is a fatal congenital disorder resulting from mutations in the myotubularin gene. Its symptoms include generalized hypertonia and muscular weakness that often leads to neonatal mortality. Animal studies show promising results about the therapeutic effects of tamoxifen on histological and pathological abnormalities associated with myotubular myopathy. Furthermore, tamoxifen has been shown to offer positive outcomes such as increased life expectancy, improvement in overall motor function, hindrance of disease progression, and prevention of lower limb paralysis [2].
2. Tamoxifen resistance
Tamoxifen has been a go-to drug for the treatment and prevention of breast cancer. However, its long-term use has developed resistance over time and it is no longer a preferred drug. Studies show that anastrozole shows more promising results and better compliance in treating breast cancer as compared to tamoxifen.
Literature suggests that the mechanism behind the resistance to tamoxifen has been associated with the excessive activation of the PI3/AKT/mTOR pathway and downregulation of estrogen receptors. Furthermore, the CD63+ CAF (cancer-associated fibroblasts) subset plays an important role in the resistance to tamoxifen. CD63+ CAF is present in the microenvironment of breast cancer cells. One study found that inhibitors of CD63+ CAF have been shown to enhance the effect of tamoxifen [3].
3. Gynecomastia
Gynecomastia is characterized by the enlargement of breast tissue. It is often caused by a disturbance in the levels of estrogen and progesterone. Tamoxifen has been used for the treatment of gynecomastia for years. A 10-year cohort study concludes that tamoxifen therapy improves symptoms of gynecomastia in every 9 out of 10 patients [4].
Furthermore, research suggests that it is a safe and effective option for pubertal gynecomastia. To achieve optimal results, tamoxifen therapy should be initiated when the disc diameter is ≥3 cm and treatment should be continued for a minimum of 6 months [5].
4. Male Infertility
Research shows that tamoxifen might improve sperm density. One study was conducted to check the effect of tamoxifen on sperm motility, morphology and density. Subjects either received 10mg tamoxifen or a placebo two times a day for three months. The researchers found that tamoxifen reduced the number of dead spermatozoa and increased sperm density as compared to the control group [6].
5. Duchenne muscular dystrophy
Duchenne muscular dystrophy (DMD) is a neuromuscular disorder that is characterized by progressive degeneration of muscles. It is often caused by a mutation in the DMD gene present on the X chromosome. Research suggests that tamoxifen is involved in the regulation of calcium homeostasis and might manage symptoms of DMD. One study in a mouse model of DMD found that tamoxifen treatment relieved motor fatigue and improved muscle strength [7].
Summary
Tamoxifen is a selective estrogen receptor modulator. It acts as an antagonist to estrogen receptors in breast tissues and reduces the proliferation of cancer cells. Research shows that it improves pathological conditions associated with myotubular myopathy and increases muscle strength in mouse models of DMD. Further, research is ongoing to confirm tamoxifen’s effect on these disorders. We don’t support its unwarranted use and offer liquid tamoxifen purchase exclusively for research and experimentation. Only buy tamoxifen if you are a licensed researcher.
References
- Farrar MC, Jacobs TF. Tamoxifen. [Updated 2023 Apr 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-.
- Gayi, E., et al., Tamoxifen prolongs survival and alleviates symptoms in mice with fatal X-linked myotubular myopathy. Nature Communications, 2018. 9(1): p. 4848.
- Gao, Y., et al., CD63+ Cancer-Associated Fibroblasts Confer Tamoxifen Resistance to Breast Cancer Cells through Exosomal miR-22. Advanced Science, 2020. 7(21): p. 2002518.
- Mannu, G.S., et al., Role of tamoxifen in idiopathic gynecomastia: A 10-year prospective cohort study. The Breast Journal, 2018. 24(6): p. 1043-1045.
- Sabancı, E., et al., Tamoxifen Treatment for Pubertal Gynecomastia: When to Start and How Long to Continue. Breast Care, 2023. 18(4): p. 249-255.
- Kotoulas, I.G., et al., Tamoxifen treatment in male infertility. I. Effect on spermatozoa. Fertil Steril, 1994. 61(5): p. 911-4.
- Nagy, S., et al., Tamoxifen in Duchenne muscular dystrophy (TAMDMD): study protocol for a multicenter, randomized, placebo-controlled, double-blind phase 3 trial. Trials, 2019. 20(1): p. 637.
What is Tamoxifen?
Tamoxifen is a medication known as a selective estrogen receptor modulator (SERM), which is frequently used to treat breast cancer that has hormone receptors. It works by blocking the estrogen receptors in breast tissue, which prevents the growth and division of cancer cells that are sensitive to hormones. To lower the chance of cancer recurrence, tamoxifen is routinely administered as adjuvant therapy after surgery or chemotherapy. In those who are at a high risk of getting breast cancer, it is also used as a preventative measure. Tamoxifen is a cornerstone in the treatment of hormone receptor-positive breast cancer due to its effectiveness in boosting rates of disease-free survival and overall survival. Researchers suggest LiquiTamo bears the potential of rectifying the condition of infertility that might have been caused by ovulatory or erectile dysfunction. This medicine acts by imposing its inhibitory action on the oestrogen receptors. Once they are bound to these receptors, there is no side left on the surface of the receptors for oestrogen to bind. The negating action of this medication can be used for reliving the malignancy of breast cancer in post-menopausal women. The absence of ovulation in such conditions can lead to problem in conceiving a baby. Lowering of estradiol in patients is followed by raise in the levels of testosterone. Tamoxifen bears immense potential that needs to be drudged with research studies which would in turn require a lot of supply of this anti-estrogenic chemical.
Tamoxifen Research
The selective estrogen receptor modulator (SERM) tamoxifen has received a great deal of attention in the field of oncology, particularly for its use in the treatment and prevention of breast cancers that express hormone receptors. Tamoxifen has been the subject of research examining its effectiveness, safety, and diverse applications in various patient populations.
Adjuvant Therapy:
Tamoxifen's use as an adjuvant therapy for hormone receptor-positive breast cancer is a prominent focus of research in this field. Its efficiency in lowering the risk of cancer recurrence following initial treatments like surgery or chemotherapy has been tested in clinical trials. Tamoxifen as adjuvant therapy dramatically increases rates of disease-free survival, overall survival, and lowers the incidence of contralateral breast cancer in both premenopausal and postmenopausal women, according to numerous studies.
Comparison Studies:
Tamoxifen research has also included comparison studies that assess its effectiveness in contrast to various treatment choices, including placebo and aromatase inhibitors. These research have looked into things such side effect profiles, overall survival, and survival without disease. Notably, studies like the ATAC and BIG 1-98 trials have shown that aromatase inhibitors are not inferior to or superior to Tamoxifen when used as adjuvant therapy in postmenopausal women.
Preventive Therapy:
Tamoxifen has been thoroughly researched for its preventative qualities in people at high risk of developing breast cancer, in addition to its use as adjuvant therapy. Tamoxifen dramatically lowers the incidence of invasive breast cancer in high-risk women, including those with a family history of the disease, according to research, including the seminal NSABP P-1 trial. For some high-risk patients, these findings have led to the suggestion of Tamoxifen as a preventive therapeutic option.
Tamoxifen Resistance:
Tamoxifen research has also focused on understanding and overcoming resistance to the drug. Studies have investigated potential biomarkers and genetic variations associated with resistance to Tamoxifen, aiming to identify patients who may benefit from alternative treatment strategies or additional interventions.
Long-term Effects and Safety:
Tamoxifen's long-term effects and safety profile have also been studied in research. Studies have looked into the dangers of using Tamoxifen, such as the risk of endometrial cancer, venous thromboembolism, and ocular toxicity. Optimizing patient care depends on recognizing these risks and putting the right monitoring and management measures in place.
Where to Buy Tamoxifen
Pinnacle Peptides offers the opportunity to buy Tamoxifen online for research purposes. As a reputable supplier, we provide high-quality Tamoxifen for sale for scientific investigations. We understand that knows how crucial it is to have trustworthy, pure research materials, therefore we make sure that researchers may get Tamoxifen of the greatest quality for their investigations. By purchasing Tamoxifen from Pinnacle Peptides, researchers gain access to a trusted source that prioritizes product quality and consistency. It is crucial to remember that Tamoxifen obtained from our online store is just meant for research and should not be utilized for human consumption or any other unlawful usage. Researchers can rely on Pinnacle Peptides to provide them with the necessary resources to advance their scientific investigations involving Tamoxifen.
Disclaimer
The products or peptides of sale we offer are intended for laboratory research use only. In purchasing any of these items, the customer acknowledges that there are risks involved with consumption or distribution of these products. These chemicals are NOT intended to use as food additives, drugs, cosmetics, household chemicals or other inappropriate applications. The listing of a material on this site does not constitute a license to its use in infringement of any patent. All of the products will be handled only by qualified and properly trained professionals. All customers represent and warrant that through their own review and study that they are fully aware and knowledgeable about the following: Government regulations regarding the use of and exposure to all products. The health and safety hazards associated with the handling of the products they purchase. The necessity of adequately warning of the health and safety hazards associated with any products. Pinnacle Peptides reserves the right to limit and/or deny sales of products to any unqualified individuals if we have reason to believe that misuse will occur.